Customer Hotline:
Customer Hotline:+1 6193853151

 Customer Hotline:
Customer Hotline:+1 6193853151

 Product Model:300 mL - 25 L.
Product Model:300 mL - 25 L.  Manufacturer nature:Manufacturer
Manufacturer nature:Manufacturer  Update time:2025-06-15
Update time:2025-06-15  Visits:240
Visits:240 product catalog
Related articles
Detailed Introduction
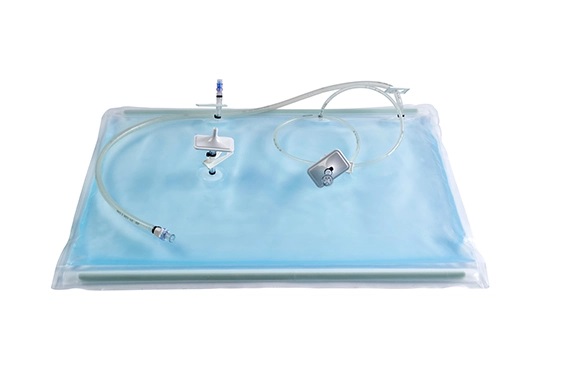
Easy use: This product is sterile for single use, providing a safe and suitable environment for cell growth, with the features of easy installation and operation.
Good stability: The bags are composed of co-extruded multi-layer films with excellent flexibility and low gas penetration rate, and are suitable for long-term cell culture.
High cell density: The perfusion function enables the high-density cell culture in a faster manner.
Good biosafety: The material-liquid contact layer is composed of EVA copolymers, which are biologically inert and can guarantee process safety.
Flexible application conditions: The bags can be used at 10-50 °C and under operating pressures up to 0.1 bar; the bags are available in various sizes to support culture volumes from 300 mL to 25 L.
Wide selection of bag type: BioLink provides cell bags for standard operation, cell therapy, and complex use; optional selections include the basic configuration, for pH & DO, perfusion, and pH & DO &perfusion.
Flexible customization of tubings, connectors, and other units to meet the needs of customers.
Complete validation documents:
*Steritity test.
*Bacterial endotoxin test.
*Integrity test.
*Extractable test.
*Chemical compatibility test.
*The biocompatibility of gamma-irradiated bags meets the following specifications:
1) ISO 10993-4: in vivo hemolysis test (extraction method).
2) USP87: Cytotoxicity test (extraction method).
3) USPClass Vl intramusclar implantation test.
4) USP88: Acute intracutaneous test.
5) USP88: Acute systemic toxicity test.