 Customer Hotline:
Customer Hotline:+1 6193853151

 Customer Hotline:
Customer Hotline:+1 6193853151

Product catalog
Related articles
Detailed Introduction
background:
Scientific research shows that more than 50% of the proteins in the human body exist in the form of glycoproteins. As one of the most important post-translational modifications of proteins in organisms, protein glycosylation has important biological functions. It not only affects the spatial conformation, biological activity, transport and positioning of proteins, but also participates in various important life activities such as molecular recognition, cell communication, cell differentiation, signal transduction, immune response, etc. In the occurrence and development of many diseases, such as tumors (most of the biomarkers approved by the FDA for the diagnosis and monitoring of cancer diseases are glycoproteins), neurodegenerative diseases, cardiovascular diseases, metabolic diseases, immune diseases and infectious diseases, the degree of protein glycosylation and abnormal sugar chain structure are accompanied . In addition, glycosylation also significantly affects the biological activity, target, transport, serum half-life, clearance rate and receptor recognition of biotherapeutics. For example, the pharmacokinetics and efficacy of recombinant erythropoietin are severely affected by its glycosylation state, and the ability of monoclonal antibodies to mediate efficacy through ADCC is affected by the fucose content in the Fc region. Therefore, protein glycosylation research is another highly promising research field in life sciences after nucleic acids and proteins, and has important theoretical and application significance. The FDA requires that all types of glycoproteins need to undergo glycoform analysis .
Peptide N-glycosidase F ( PNGase F, EC 3.5.1.52) is an endoglycosidase that specifically removes N-linked glycans from glycoproteins. It is an important tool enzyme for glycosylation research and is widely used in the study of N- linked glycan sites, structural information and functional relationships on therapeutic proteins. Rhinogen ® PNGase F (Glycerol-free) is a recombinant PNGase F expressed in Escherichia coli BL21 . It can completely release most N-glycans (except for core fucose containing α-1,3 linkages, which are common in plant and insect glycoproteins) from the protein molecules to which they are connected. It can cut between the innermost N-acetylglucosamine (GlcNAc ) and asparagine residues ( Asn ) of the high mannose, hybrid and complex oligosaccharide parts of N-linked glycopeptides or glycoproteins to release complete oligosaccharide chains, as shown in Figure 1A. While releasing the sugar chains , the asparagine residues on the amino acid characteristic sequence are converted to aspartic acid residues, which give the protein a + 1Da mass change, as shown in Figure 1B. It is highly active against a variety of substrates, including purified glycoproteins and complex mixtures of human serum glycoproteins. In addition, the absence of glycerol makes the enzyme compatible with HPLC and mass spectrometry workflow environments under non-denaturing conditions. Phosphate, sulfate, and sialic acid groups attached to N-linked oligosaccharides do not affect the release of N-glycans .
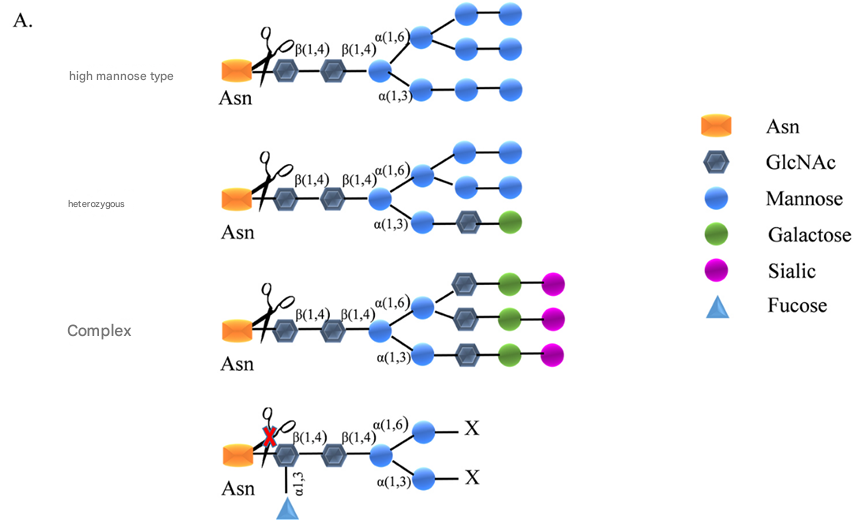
Figure 1A. PNGase F cleavage site





