 Customer Hotline:
Customer Hotline:+1 6193853151

 Customer Hotline:
Customer Hotline:+1 6193853151

Product catalog
Related articles
Detailed Introduction
background:
Rhinogen ® Protein Deglycosylation KitⅠ(for O -linked Glycans) contains O -Glycosidase and α2-3,6,8,9 Neuraminidase, both of which are important tool enzymes for glycosylation research. The combination is used for the enzymatic cleavage and release of glycoprotein O - linked sugar chains . O -Glycosidase can release the O -linked disaccharides of Core 1 (Gal-β(1-3)-GalNAc-) and Core 3 ( GlcNAc -β(1-3)-GalNAc-) linked to the hydroxyl groups of Ser or Thr residues in glycoproteins, as shown in Figure 1. Any modification of the core structure can block the action of O -Glycosidase, and the most common modifications are mono-, di-, or tri-sialylation of the core structure. Rhinogen ® α2-3,6,8,9 Neuraminidase can release the non-reducing terminal α (2,3)-, α (2,6)-, α (2,8)-, and α (2,9)-sialic acid residues of O -linked sugar chains, as shown in Figure 2. The release rates of different sialic acid residues are roughly α (2,6)->α (2,3)->α (2,8)->α (2,9)-. This combination mode of sialidase allows for more complete enzymatic cleavage and release of O - linked sugar chains . All enzymes and reagents included in this kit are compatible with downstream HPLC and MS.
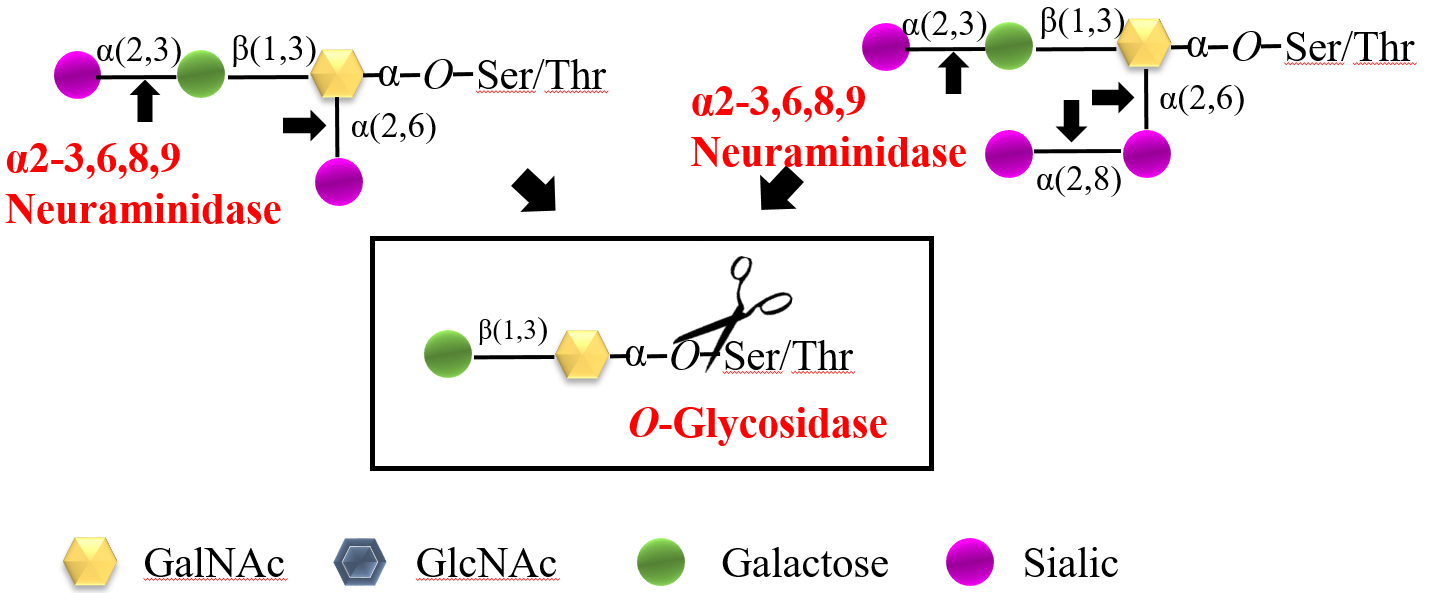
Figure 1. O -Glycosidase and α2-3,6,8,9 Neuraminidase are used to release O - linked sugar chains from glycoproteins by enzymatic cleavage.
Figure 2. Rhinogen ® α2-3,6,8,9 Neuraminidase action site
Packing Specifications:
Rhinogen ® Protein Deglycosylation KitⅠ(for O -linked Glycans) packaging specifications are as follows:
|
Catalog Number |
composition |
Specification |
volume |
|
QPF-008 |
O -Glycosidase α2-3,6,8,9Neuraminidase |
1,200,000U 0.6U |
30μl 30μl |
Supporting reagents:
The supporting reagents provided by Rhinogen ® Protein Deglycosylation KitⅠ(for O -linked Glycans) are as follows:
|
Reagents |
Element |
|
10× Denaturing Buffer |
5% SDS, 0.4M DTT |
|
10× Glyco Buffer 2 |
0.5MSodium Phosphate(pH7.5 at 25°C) |
|
10% NP-40 solution |
10%NP-40 |
Product Source:
All enzymes in this kit are recombinant enzymes expressed in E. coli BL21 and isolated and purified: O -Glycosidase has a molecular weight of 147kDa; α2-3,6,8,9 Neuraminidase has a molecular weight of 66KDa.
Product quality:
SDS-PAGE analysis showed that the purity was ≥95%; no contaminating exoglycosidase, endoglycosidase and protease activity was detected.
Enzyme activity definition :
1 unit of O -Glycosidase activity is defined as the amount of enzyme required to catalyze the release of 0.68 nmol of O - linked disaccharide from 5 mg of non-denatured fetuin digested with sialidase in 1 hour at 37°C and pH 7.5 in a 100 μl reaction system . O -Glycosidase =1 unit NEB O -Glycosidase.
1 unit of α2-3,6,8,9 Neuraminidase activity is defined as the amount of enzyme required to catalyze the release of 1 μmol of p-nitrophenol in 1 minute using p-nitrophenyl-α-DN-acetylneuraminic acid as substrate at 37°C and pH 5.5. 1U Rhinogen ® α2-3,6,8,9 Neuraminidase = 1 U Prozyme Glyko ® Sialidase ATM.
Storage conditions:
-20°C.
Features:
Rhinogen ® Protein Deglycosylation KitⅠ(for O -linked Glycans) has the characteristics of high stability and high specific activity. It is a combination of two highly purified and very stable glycosidases, suitable for the effective release of common O -linked glycans on glycoproteins in proteomics and glycobiology research .
1. High purity: no contaminating protease/other glycosidase, purity ≥95%;
2. High stability: Enzymes and reagents in each batch of Kit are strictly quality controlled to achieve high stability;
3. High specific activity: effectively release sialic acid-modified O -linked glycans on glycoproteins ;
4. HPLC and MS compatible: All enzymes and reagents in the kit are compatible with downstream HPLC and MS.
application:
1. Glycan structure analysis;
2. Binding site and function analysis;
3. Characterization and quality control of therapeutic glycoproteins;
4. Proteomic analysis to eliminate the heterogeneity of glycoproteins.
Recommended use:
Deglycosylation protocol under denaturing conditions:
1. Take 10-100µg of glycoprotein solution, add 1µl 10× Denaturing buffer, and add purified water to make the reaction system 10µl;
2. Treat the above 10µl system at 100℃ for 10min to completely denature the glycoprotein;
3. Cool at room temperature for 5 minutes;
Glyco buffer 2, 2µl 10% NP-40 solution, 1-4µl O- Glycosidase, 1-2µl α2-3.6.8.9Neuraminidase to the above denaturation system, add purified water to a total reaction volume of 50µl, and mix gently;
5. Incubate at 37°C for 1-5 hours;
Note: For different glycoprotein samples, it is necessary to experiment to find the optimal enzyme concentration and reaction time.
Deglycosylation protocol under native conditions:
1. Take 10-100µg of glycoprotein substrate solution, add 2µl 10× Glyco buffer 2, purified water, 1-4μl O -Glycosidase, 1-2μl α2-3.6.8.9Neuraminidase, so that the total reaction volume is 20µl, and mix gently;
2. Incubate at 37°C for 1-4 hours.
Note: Generally speaking, whether the glycoprotein is denatured or not does not significantly affect the efficiency of O - linked sugar chain removal, but this does not rule out individual cases. It is recommended to choose an appropriate method based on the characteristics of your own substrate.
When deglycosylation of native glycoproteins , it is recommended to denature an equal amount of glycoprotein sample and then perform enzyme digestion simultaneously as a positive control to determine the extent of the deglycosylation reaction under non-denaturing conditions.
Instructions:
1. The above operation method is intended to be a general guide for the operation of Rhinogen ® Protein Deglycosylation KitⅠ(for O -linked Glycans) as a deglycosylation reagent. For different glycoprotein samples, the deglycosylation activity is highly dependent on the reaction conditions. It is recommended to perform appropriate optimization or determine the optimal operation method based on experience.
2. The deglycosylation system can be scaled up or down linearly;
3. Since the denaturation buffer contains SDS, SDS will inhibit Rhinogen ® The activity of O -Glycosidase is inhibited, so NP-40 with a final concentration of 1% needs to be added to the denatured glycoprotein reaction system, which can effectively relieve the inhibition of SDS on the activity of O -Glycosidase ;
4. This product is suitable for natural or denatured glycoproteins. For the deglycosylation of natural glycoproteins , more enzymes and longer reaction time may be required;
5. EDTA, p-chloropropylbenzenesulfonic acid, Mn 2+ and Zn 2+ have a certain inhibitory effect on the activity of O -Glycosidase . Under the condition of 1mM of each substance, the enzyme activity recovery rates are 63%, 60%, 44% and 66% respectively;
6. This product is for research use only and is not intended for human or animal diagnosis or treatment.
Frequently asked questions
Q1: Is this kit compatible with downstream HPLC and MS?
A1: Yes, all enzymes and buffers in the Rhinogen ® Protein Deglycosylation Kit Ⅰ (for O -linked Glycans) are compatible with downstream HPLC and MS applications. Proteins can be prepared for HPLC and MS analysis by microdialysis or microfiltration.
Q2: After treating glycoprotein with O Glycosidase, no sugar chains were removed. What could be the reason?
A2: Protein glycosylation modification includes N-glycosylation and O-glycosylation. O-Glycosidase is suitable for releasing the O-linked disaccharide cores of Core 1 and Core 3 attached to Ser/ Thr . If the substrate is confirmed to contain O-glycans, please make sure to use Neuraminidase at the same time to release the terminal sialic acid modification group. At the same time, due to the steric hindrance effect (secondary and tertiary structures of proteins) that can hinder endoglycosidase from reaching its substrate action site, denaturation before deglycosylation helps to effectively release O-linked glycans. If you do not want to denature, consider adding more enzyme or extending the incubation time.
Q3: How much Rhinogen ® O Glycosidase is needed to release the sugar chains of glycoproteins under native conditions ?
A3: When the protein is not denatured, O-Glycosidase may have difficulty reaching the cleavage site of the sugar chain due to steric hindrance (due to secondary and tertiary protein structures). Adding more enzyme and extending the reaction time may help improve the efficiency of sugar chain release , but for different glycoprotein samples, the deglycosylation activity is highly dependent on the reaction conditions. It is recommended to perform appropriate optimization or determine the optimal operation method based on experience.
Q4: Can PNGase F, O Glycosidase and α 2-3,6,8,9 Neuraminidase be used at the same time?
A4: Yes. Rhinogen ® PNGase F and O-Glycosidase use the same buffer and reaction conditions, while α2-3,6,8,9 Neuraminidase has a very wide pH range (pH4.5-8.5) and has good activity in the PNGase F and O-Glycosidase reaction system. The three can be used simultaneously.