 Customer Hotline:
Customer Hotline:+1 6193853151

 Customer Hotline:
Customer Hotline:+1 6193853151

Product catalog
Related articles
Detailed Introduction
background:
O -Glycosidase ( EC 3.2.1.97) is an important enzyme for glycosylation research. Rhinogen ® O -Glycosidase is a recombinant enzyme expressed in Escherichia coli BL21 with the O -Glycosidase gene recombined. It can release the O-linked disaccharides of Core 1 (Gal-β(1-3)-GalNAc-) and Core 3 ( GlcNAc -β(1-3)-GalNAc-) linked to the hydroxyl groups of Ser or Thr residues in glycoproteins, as shown in Figure 1. The enzyme is specific for α-GalNAc binding, but has no obvious preference for Ser or Thr residues. At the same time, the denaturation of glycoproteins does not significantly affect the efficiency of O - deglycosylation . Any modification of the core structure can block the action of O-Glycosidase. The most common modification is mono-, di- or tri-sialylation of the core structure. In addition, the core structure may be further substituted and modified by fucose, N-acetylglucosamine or N-acetylgalactosamine residues. Monosaccharides outside the core structure must be cut in sequence by a series of exoglycosidases until only the Core 1 or Core 3 disaccharide core remains. Then O -Glycosidase can completely release the disaccharide core from the Ser or Thr residue, as shown in Figure 2. Usually, the use of O -Glycosidase requires the use of sialidase.
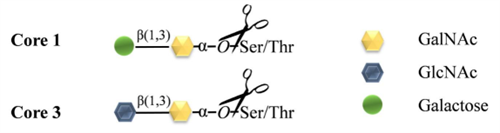
Figure 1 Types of O -linked disaccharide cores that Rhinogen ® O -Glycosidase acts on





