 Customer Hotline:
Customer Hotline:+1 6193853151

 Customer Hotline:
Customer Hotline:+1 6193853151

Product catalog
Related articles
Detailed Introduction
background:
Among clinical infusion reactions, pyrogenic reactions are the most harmful and have the highest incidence. Endotoxin (i.e., lipopolysaccharide molecules in the cell wall of Gram-negative bacteria, LPS) is the most thoroughly studied and most common biological pyrogen. Even if pg -level endotoxin is injected, it can cause severe pyrogenic reactions in patients, causing fever, shock, and even death. Endotoxins are ubiquitous and difficult to inactivate, which is a challenge for the pharmaceutical and medical device industries. Therefore, sensitive and reliable endotoxin analysis technology is very necessary.
Since the 1970s, the horseshoe crab test for endotoxin detection has been used in the medical field and has been widely adopted by countries around the world and has been made a legal bacterial endotoxin test method. Traditional horseshoe crab reagents are divided into two types: gel method and photometric method, and the photometric method is divided into turbidity method and colorimetric method. Among them, the dynamic colorimetric method has the widest detection sensitivity range, can quantitatively detect endotoxin content, and can provide data audit tracking records.
In the presence of endotoxin, the lysate of the dynamic colorimetric horseshoe crab reagent will begin to cleave the chromogenic substrate, causing the solution to turn yellow. The time required for the solution to change color is inversely proportional to the amount of endotoxin present. The concentration of the unknown sample can then be calculated from the standard curve. However, as a traditional horseshoe crab reagent, the dynamic colorimetric horseshoe crab reagent still has disadvantages that cannot be avoided due to the production raw materials. For example, the problem of nonspecific interference: in addition to reacting with endotoxin, the horseshoe crab reagent will also react with (1-3)-β-D-glucan, resulting in false positive results. Another example is the batch stability problem: the horseshoe crab reagent uses horseshoe crab blood as the production raw material. Due to seasonal and regional differences, batch differences in horseshoe crab reagents have become a common phenomenon.
With the advancement of biotechnology, recombinant protein production technology has gradually replaced natural extraction technology. In order to protect horseshoe crab resources and eliminate the disadvantages of traditional horseshoe crab reagents such as nonspecific interference caused by bypass factor G , it is the current development trend to develop a kit that does not rely on animal horseshoe crab sources and can take into account high sensitivity and quantitative detection of endotoxins through recombinant expression.
Overview:
Rhinogen® Recombinant Cascade Reagent ( rCR ) uses genetic recombination technology to express Factor C, Factor B and proclotting enzyme in horseshoe crab blood cells; when the recombinant factor C binds to endotoxin and is activated , it activates the recombinant factor B, which then activates the recombinant proclotting enzyme, turning it into a biologically active coagulase. Finally, the coagulase recognizes and catalyzes the downstream substrate with a chromogenic group to produce a color reaction. The intensity of the color reaction is positively correlated with the endotoxin concentration, thereby quantitatively detecting endotoxin.
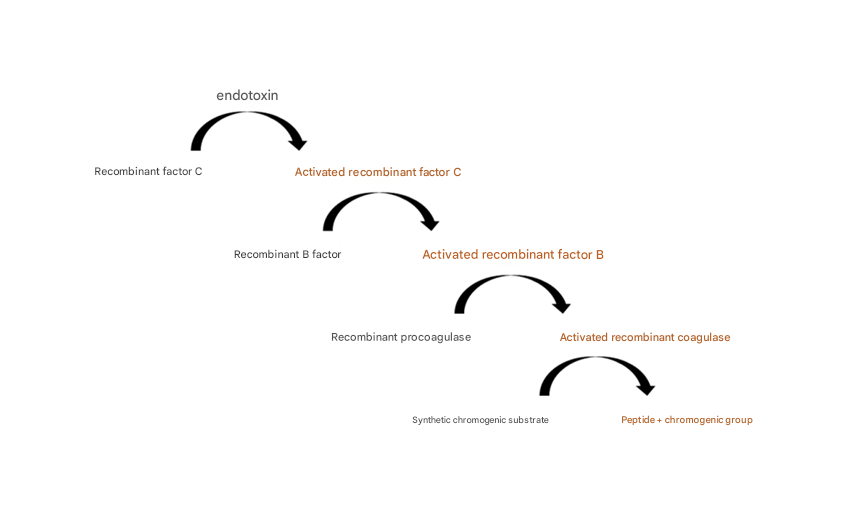
Figure 1. Reaction principle of Rhinogen® recombinant horseshoe crab reagent endotoxin detection kit
Reagent packaging:
the Rhinogen ® recombinant horseshoe crab reagent endotoxin test kit are as follows:
|
Kit Components |
Part Number |
Specification |
|
RAF-03 |
4×28T |
|
|
Recombinant horseshoe crab reagent lyophilized powder |
RAF-03A |
4vials |
|
Reconstitution Buffer |
RAF-03B |
4vials |
|
Pyrogen-free water |
RAF-03C |
2vials |
|
Bacterial Endotoxin Working Standard |
RAF-03D |
2vials |
Product Features:
Rhinogen® Recombinant Horseshoe Crab Reagent Endotoxin Test Kit is an endotoxin test product that does not rely on animal-derived ingredients, has high sensitivity, high specificity, and can be stably and continuously provided. It has the following characteristics:
1. The operation process is consistent with
the dynamic colorimetric endotoxin detection reagent, the detection equipment
is the same, and the analysis method is the same;
2. Convenient detection, single- step reconstitution , and the reagent dosage
is 100μl;
3. The detection time is only 90-120 minutes;
4. Detection in 96-well plates, easy to operate and high throughput;
5. The sensitivity range is 0.005EU/ml~5EU/ml;
6. Endotoxin specificity, no G factor bypass interference, avoiding false positive results;
7. It eliminates the reliance on animal-derived reagents and complies with the 3R substitution principle, providing higher supply security;
8. Recombinant expression production, good product batch consistency ;
9. This kit can be used for endotoxin detection of raw materials, auxiliary materials, intermediate products and released products of injectable drugs for human and animal use (such as chemicals, radiopharmaceuticals, antibiotics, biological products, etc.) and medical devices (such as dialysate, implantable devices, etc.).
Storage conditions:
Transported in ice packs. After receiving the kit, please store it at 2℃~8℃ immediately.