 Customer Hotline:
Customer Hotline:+1 6193853151

 Customer Hotline:
Customer Hotline:+1 6193853151

Product catalog
Related articles
Detailed Introduction
Product Introduction
background:
Among clinical infusion reactions, pyrogenic reactions are the most harmful and have the highest incidence. Endotoxin (i.e., lipopolysaccharide molecules in the cell walls of Gram-negative bacteria, LPS) is the most thoroughly studied and most common biological pyrogen. Injection of even trace amounts of LPS at the pg level can cause severe pyrogenic reactions in patients, causing fever, shock, and even death. Endotoxins are ubiquitous and difficult to inactivate, which is a challenge for the pharmaceutical and medical device industries. Therefore, sensitive and reliable endotoxin analysis technology is very necessary.
The amoebocytes of horseshoe crabs are highly sensitive to LPS. The endotoxin detection method of horseshoe crab reagents made from the lysate of horseshoe crab blood amoebocytes has been used in the medical field since the mid-1970s and was soon widely adopted by countries around the world. The Chinese Pharmacopoeia and the European and American Pharmacopoeia have designated it as a legal bacterial endotoxin test method. After decades of development and improvement, the endotoxin detection method of horseshoe crab reagents has been applied to pharmaceuticals, medical devices, non-oral preparations, pharmaceutical products, biological preparations, water quality, food testing, and scientific research.
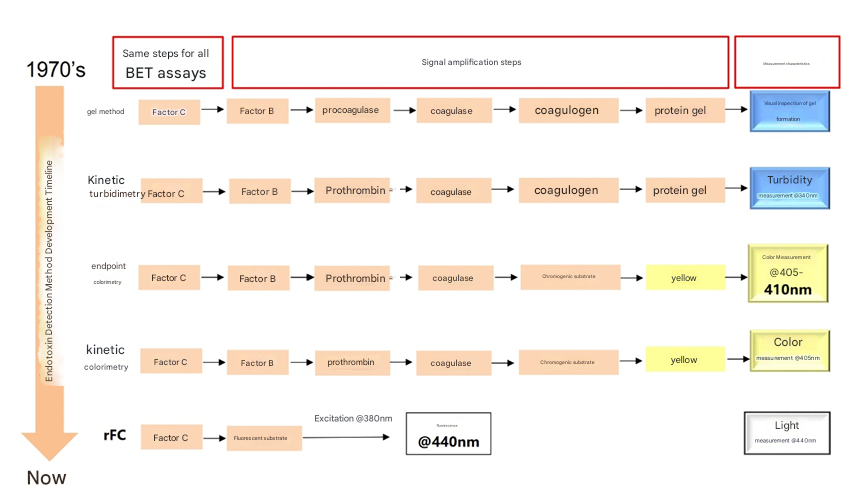
Figure 1. Development and improvement history of the horseshoe crab test method
crab blood cell lysate with complex components as the reaction raw material has insurmountable defects. For example, the problem of nonspecific interference: horseshoe crab reagents can react with (1-3)-β-D-glucan in addition to endotoxins, resulting in false positive results. Another example is the batch stability problem: horseshoe crab reagents use horseshoe crab blood as the production raw material. Due to seasonal and regional differences, batch differences in horseshoe crab reagents have become a common phenomenon. Finally, environmental pollution and other reasons have caused a decrease in the number of horseshoe crabs in the ocean , making it increasingly difficult to obtain raw materials. With the rapid growth in demand for human and animal injectable drugs (such as chemicals, radiopharmaceuticals, antibiotics, biological products, etc.) and medical devices (such as dialysate, implantable devices, etc.), it is becoming increasingly important to develop fast and reliable alternative methods for quantitative detection of endotoxins.
Overview:
Horseshoe crab factor C is a serine protease that is sensitive to bacterial endotoxins in horseshoe crab blood cells. In the horseshoe crab blood coagulation system reaction mediated by bacterial endotoxins, horseshoe crab factor C is the first to be activated by endotoxins and initiates the blood coagulation cascade reaction in the entire horseshoe crab blood cells. Rhinogen ® recombinant factor C is a genetically recombinant expression of the eastern horseshoe crab ( Tachypleus tridentatus ) Recombinant Factor C ( rFC ), which is activated by endotoxin, can cleave the substrate to obtain free fluorescent groups. The release of fluorescent groups is proportional to the concentration of endotoxin, so that endotoxin can be quantitatively detected. Compared with the classic horseshoe crab reagent endotoxin detection method, the recombinant factor C endotoxin detection method has higher specificity, better specificity, precision, accuracy, linear range and quantitative limit, and is an improved method of the current horseshoe crab reagent endotoxin detection method.
Product Packaging:
This kit contains enough reagents to perform 96 tests (96-well plate: 100 μl/well/test):
|
name |
Part Number |
Specification |
|
Rhinogen ® Recombinant Factor C Endotoxin Detection Kit |
RAF-01 |
96 tests |
|
Kit Components: |
||
|
• Endotoxin working standard |
100EU/bottle×1 |
|
|
• Fluorescent substrate reagents |
6ml/bottle×1 |
|
|
• Assay buffer |
5ml/bottle×1 |
|
|
• rFC enzyme solution |
0.6ml/bottle×2 |
|
|
• Pyrogen-free water |
30ml/bottle×1 |
|
Storage conditions:
Transported in ice packs. After receiving the kit, please store it immediately at 2~8℃. The shelf life of unopened products is 12 months. After dissolving, the endotoxin working standard is stored at 2~8℃ and is valid for two weeks.
Product Features:
Rhinogen ® Recombinant Factor C Endotoxin Assay Kit is an endotoxin assay reagent that does not rely on animal-derived components, has high sensitivity, high specificity, and can be stably and continuously provided as an alternative to the horseshoe crab reagent. It has the following characteristics:
1. Endpoint fluorescence measurement, comparable to other quantitative LAL methods
2. Liquid reagents are easy to use
3. Sensitivity range from 0.005 to 5EU/ml
4. Endotoxin specificity, no G factor bypass interference
5. Eliminates the reliance on animal-derived reagents and complies with the 3R substitution principle
6. Recombinant expression production, good product batch consistency
7. No reliance on animal-derived ingredients, providing greater supply security
8. This kit can replace the traditional horseshoe crab reagent for the endotoxin detection of raw materials, auxiliary materials, intermediate products and released products of human and animal injectable drugs (such as chemicals, radiopharmaceuticals, antibiotics, biological products, etc.) and medical devices (such as dialysate, implantable devices, etc.).
Frequently asked questions
Q1: No signal in sample detection
A1:
|
reason |
Solution |
|
Pipetting errors |
Retest |
|
Interference |
The spike pre-experiment or the spike recovery rate of each experiment is unqualified, indicating interference. Dilute the sample or eliminate the interference. |
|
Inappropriate pH |
Check the pH value of the sample and adjust it to the neutral range of 6-8 |
Q2: Low signal strength
A2:
|
reason |
Solution |
|
Instrument sensitivity (gain) is too low |
To increase sensitivity, higher gain is required |
|
The microplate reader detection parameters are set incorrectly (e.g., optics) |
Check the running instrument setting parameters |
|
Incubation temperature is too high/too low |
Check and calibrate temperature |
|
Reagent failure (transportation or storage) or expiration |
Check storage conditions and packaging materials; contact technical service to use new kit or new reagents |
High background signal in standard and negative control wells
A3:
|
reason |
Solution |
|
LPS contamination assay components (substrate reagent, assay buffer or rFC enzyme solution) |
When using newly opened reagents, be sure to use sterile, pyrogen-free tips |
Q4: Linearity of standard curve R 2 <0.98
A4:
|
reason |
Solution |
|
Endotoxin working standard solution dilution error |
Re-dilution accurately |
|
The low sensitivity of the fluorescence detection of the microplate reader causes the fluorescence value at 5EU/ml to exceed the instrument's detection range |
Use a microplate reader with high fluorescence detection sensitivity |